NRF2 in COST Action 20121
Since the start of BenBedPhar in November 2021, I would dare to say that our activity has been more than intense. After signing with COST the economical agreement for the first year, in just three months that included winter holidays, we have built the BenBedPhar organizational structures: five Working Groups, Grant Award Committee, Scientific Communication Coordinator, etc, that have already started their specific activities.
We have organized the public section of BenBedPhar’s webpage and a private section organized in six Slacks, one for each working group and one for reports. We already had two online meetings of the Management Committee and one of the Core Group.
We are particularly proud of the BenBedPhar Marathon that was held online in January 10-11, with 45 communications and over 100 participants.
At this time, BenBedPhar members account for over 170 participants from 31 EU countries, Japan and USA . These figures are large considering that our network is at the very beginning.
Life is movement and we keep moving … So, visit our webpage to get information about special publishing issues on NRF2 that we are organizing in Antioxidants and in Free Radical Biology & Medicine. Have a look at the webinars in collaboration with Sapienza University of Rome, starting next month, or surf along our first Newsletter. Here you will find educational comments on molecular biology, pharmacology and medicine of NRF2, as well as the “Hot from Pubmed” section on some relevant articles published during the last three months.
Enjoy!
Antonio Cuadrado
Chair of COST Action 20121, BenBedPhar
Autonomous University of Madrid
Comments from the Working Groups
That’s quite a story!
The hypomorphic KEAP1 resolves the bird paradox
Birds have a high oxygen consumption rate due to metabolic demand associated with flying which consumes ten to twenty times more oxygen than resting. Yet, they manage oxidative stress burden very effectively. This “bird paradox” (Satoh Trends Endocrinol Metab. 2021) can be explained by constitutive NRF2 activity counteracting reactive oxygen species (ROS) leakage and oxidative damage.
Gianni M. Castiglione et al. showed recently that intrachromosomal rearrangement in Neoaves led to the loss of KEAP1 sequence coding C-terminal amino acids (Castiglione et al. Nat Commun. 2020). The lost fragment of KEAP1 corresponds to the part of the Kelch-repeat domain, which binds DLG and ETGE motifs of NRF2. Consistent with this, NRF2 repression by KEAP1 is lost, and NRF2 is hyperactivated in Neoaves.
Such adaptation provides essential antioxidant protection for this clade. Since KEAP1 is highly conserved in the basal Avians: the Palaeognathae (i.e. kiwi) and the Galloanserae (i.e. chicken), but not in Neoaves, partial KEAP1 deletion in the evolution of birds may have occurred during the early Tertiary period (Satoh Trends Endocrinol Metab. 2021).

Studies in Keap1 knockout mice showed that constitutive activation of NRF2 is lethal postnatally. Keap1 KO mice present severe growth retardation and die within 21 days after birth, most likely due to starvation caused by hyperkeratosis of the digestive tract (Wakabayashi et al. Nat Genet. 2003). These two opposite phenotypes of overactive NRF2 -lethal in mice and harmless in birds- are a thought-provoking divergence. They may be explained by the loss of ARE elements in the promoter regions of β-keratin in Neoaves.
Similar aberrations were found in the neoavian glutathione S-transferase A2 (GSTA2) gene, whereas the NQO1 locus was shown to be deleted entirely. As the authors conclude, these compensatory mutations may have been required for the loss of KEAP1 repressive function to be evolutionarily successful (Castiglione et al. Nat Commun. 2020).
Anna Grochot-Przęczek
WG1 leader
Jagiellonian University. Poland
Glued forever?
On the NRF2 regulation by KEAP1 and the effect of electrophilic NRF2 activators
A number of models describing the regulation of NRF2 by KEAP1 and the effect of electrophiles on the KEAP1/NRF2 protein-protein interactions have been proposed; these are reviewed in (Baird and Dinkova-Kostova, 2011). One of the earliest models, named “Sequester and Release” postulated that under homeostatic conditions, dimeric KEAP1 sequesters NRF2 in the cytoplasm by binding to the actin cytoskeleton. Electrophiles react with specific cysteine residues in KEAP1, leading to release of NRF2, allowing the transcription factor to translocate to the nucleus, heterodimerize with a small MAF transcription factor, and induce the expression of NRF2-target genes. This logical model was in line with a common paradigm in biology, where a transcription factor is bound to a cytoplasmic repressor until a specific signal triggers its release.
Whereas it was a valuable working model at the time and some of its aspects are correct, subsequent research suggested the need for revision, and it has now been established that cysteine modifications within KEAP1 by electrophiles do not cause release of NRF2.
Unfortunately, the “Sequester and Release” model is still being used by many researchers and educators, and is present in the published literature.
Following the discovery of the high-affinity ETGE and the low-affinity DLG motifs, thorough which NRF2 binds to each of the Kelch domains of KEAP1, the ‘‘Two-Site Substrate Recognition’’ or ‘‘Hinge & Latch’’ model was proposed, whereby binding of these two motifs of NRF2 to the KEAP1 homodimer is an obligatory requirement for ubiquitination and proteasomal degradation of the transcription factor (McMahon et al., 2006; Tong et al., 2006). NRF2 activators cause dissociation of the DLG-binding motif of NRF2 from KEAP1 as a latch, while the ETGE motif of NRF2 remains attached to KEAP1 as a hinge. Recent titration nuclear magnetic resonance (NMR) spectroscopy experiments demonstrated that this mechanism is employed by KEAP1-NRF2 protein-protein interaction (PPI) inhibitors, but not by electrophilic NRF2 inducers (Horie et al., 2021). Overall, the current experimental evidence supports the model depicted in Figure 2 (Dinkova-Kostova and Kostov, 2012), whereby, at homeostatic conditions, NRF2 binds through its ETGE and DLG motifs, to the Kelch domains of the KEAP1 dimer and is continuously targeted for ubiquitination and subsequent proteasomal degradation. Electrophilic NRF2 activators bind covalently to specific cysteine sensors of KEAP1, leading to loss of its ability to target NRF2 for ubiquitination.
Consequently, the binding sites within the Kelch domains of the KEAP1 dimer are saturated with unmodified NRF2, and the newly synthesized NRF2 accumulates, heterodimerizes with a small MAF transcription factor, and the complex binds to antioxidant response elements (AREs) in the promoter regions of NRF2-target genes, inducing their transcription.
Albena Dinkova-Kostova
WG2 leader
University of Dundee, UK
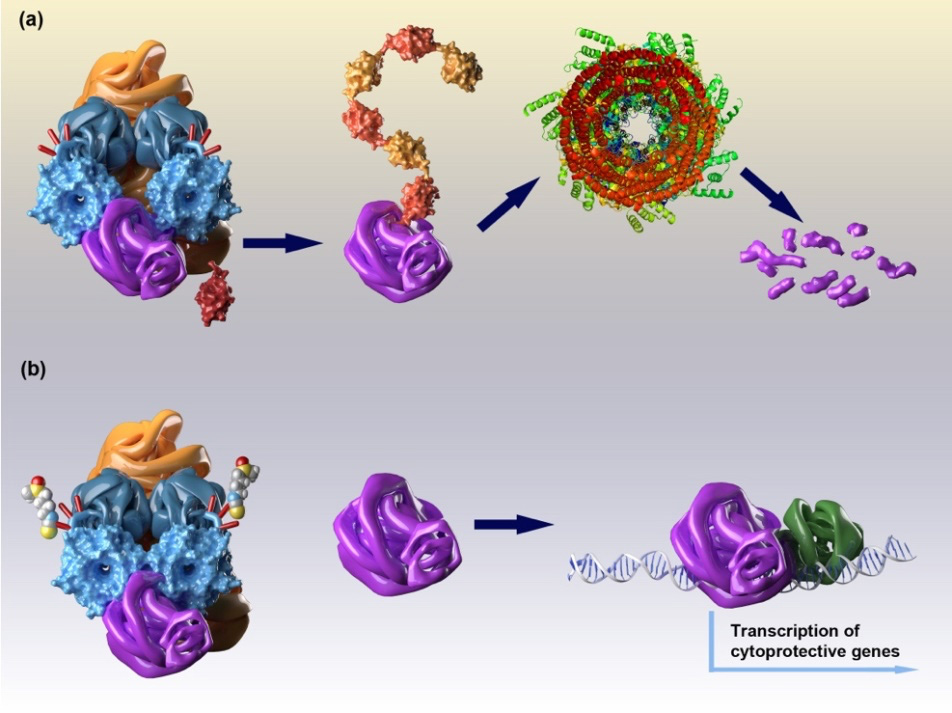
Figure 2. Current model of NRF2 regulation by KEAP1 and the effect of electrophilic NRF2 activators. (a) At homeostatic state, NRF2 (purple) binds to the Kelch domains of KEAP1 (blue) and is ubiquitinated via the association of KEAP1 with Cullin 3-based E3 ubiquitin ligase (orange). Polyubiquitinated NRF2 (the ubiquitin molecules are shown as alternating dark red and orange) enters the proteasome, where it is degraded. (b) Electrophilic NRF2 activators, such as sulforaphane (shown as space-filling model), bind covalently to specific cysteine sensors (red sticks) of KEAP1, leading to loss of its substrate adaptor function. Consequently, KEAP1 is saturated with unmodified NRF2, and the newly synthesized NRF2 accumulates, heterodimerizes with a small MAF (green), and the complex binds to antioxidant response elements (AREs), inducing NRF2-target gene transcription. Figure from: Dinkova-Kostova and Kostov, 2012.
Fighting AGEs for healthy AGIng
NRF2 activates dicarbonyl detoxification
Advanced glycation end products (AGEs) are toxic metabolites that derive in part from reactions of proteins or DNA with dicarbonyl compounds, such as methylglyoxal (MG). MG-adducts compromise the function for these biomolecules and participate in organ damage. A well-establish function of AGEs is the induction of inflammatory responses, at least in part through the activation for their cognate receptor (RAGE), leading to chronic low grade inflammation. Elevated levels of MG and MG-adducts are found during ageing and in chronic diseases and participate in endothelial and vascular dysfunction, type 2 diabetes mellitus, atherosclerosis, neurodegeneration or cardiovascular disease.
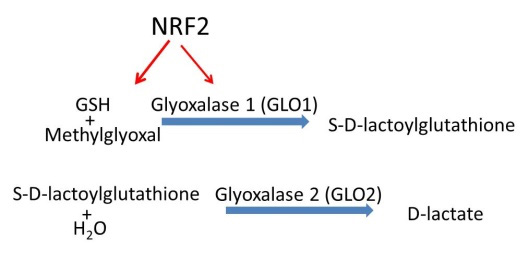
Figure 3. Detoxification of methylglioxal by the glioxalase system. This system is form by glyoxalase 1 (GLO1), glyoxalase 2 (GLO2), and glutathione (GSH). NRF2 activates this system by inducing the expression of genes involved in GSH production and recycling as well as up-regulation of the GLO1 coding gene.
MG is detoxified by the enzyme Glyoxalase 1 (GLO1), using reduced glutathione (GSH) as a co-factor. Xue et al., identified a functional NRF2-regulated antioxidant-response element in the 5′-untranslated region of exon 1 of the GLO1 coding gene. Moreover, NRF2-knockout mice show decreased GLO1 mRNA and protein levels in several organs. Therefore, through direct up-regulation of GLO1 and of genes involved in GSH production, NRF2 may be instrumental in MG detoxification and preventing low-grade chronic inflammation originating from dicarbonyl toxicity.
There is an active search for biomarkers that could be used in monitoring the response to NRF2 activators in humans. At least in the case of sulforaphane, it is possible to detect decreases in cellular and extracellular concentrations of MG and MG-derived protein adducts (Alfarano et al., 2018). Therefore, a very promising yet little explored possibility to monitor the basal status of the NRF2 system or the response to NRF2 activators may be the analysis of MG-derivate levels.
References:
– Xue et al. Transcriptional control of glyoxalase 1 by Nrf2 provides a stress-responsive defence against dicarbonyl glycation. Biochem J. 2012, 443:213-22.
– Alfarano et al. The Effect of Sulforaphane on Glyoxalase I Expression and Activity in Peripheral Blood Mononuclear Cells. Nutrients.
Antonio Cuadrado
WG3 member
Autonomous University of Madrid
Brigitte Winklhofer-Roob
WG3 Co-Leader
Karl-Franzens Universität Graz. Austria
Hope for Friedreich Ataxia
NRF2 promising technology
Friedreich’s ataxia (FA) is a rare degenerative autosomal recessive hereditary neuromuscular disorder with no approved therapies. Reata Pharmaceuticals, a clinical-stage company, had recently completed a pre-NDA meeting with FDA and plan to submit Omaveloxolone, an oral NRF2 activator, in the first quarter of 2022. The company had recently evaluated clinically the safety and efficacy of Omaveloxolone in patients with FA (Phase II) and found that it was well tolerated by the patients and could significantly improve neurological function. It should be noted that Omaveloxolone has been granted Orphan Drug designation for Friedreich’s ataxia treatment from both the European Commission and FDA, and therefore the company is eligible to receive regulatory assistance as well as extended marketing exclusivity.

Figure 4. Unknown Author is licensed under CC BY-SA-NC
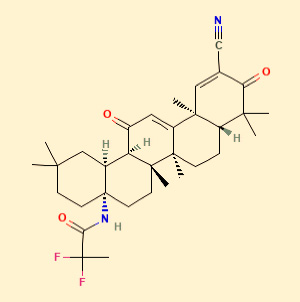
Figure 5. Omaveloxolone, PubChem
Omaveloxolone (N-(2-cyano-3,12-dioxo-28-noroleana-1,9(11)-dien-17-yl)-2-2-difluoropropanamide) is a synthetic oleanane triterpenoid that activates NRF2 in numerous pre-clinical models. The molecule harness both the anti-inflammatory and antioxidative activity of NRF2. The company has an additional NRF2 activator in their pipeline, Bardoxolone methyl, which is tested in chronic kidney disease.
In other news, Calithera Biosciences has recently communicated that the Phase II KEAPSAKE clinical trial in stage IV non-squamous non-small cell lung cancer was suspended due to a lack of clinical benefit. The study, investigating combined therapy of telaglenastat (glutaminase inhibitor) with pembrolizumab and chemotherapy, was conducted in patients with KEAP1/NRF2-mutated. In this sub-population, NRF2 aberrant activation confers to cancer cells high-level resistance to anticancer drugs as well as protection from microenvironmental stress. The negative results clearly demonstrate the need to develop safe and clinically effective NRF2 inhibitors.
Guy Cohen
WG4 leader
The Dead-Sea & Arava Science Center, Israel
The end of medicine as we know it
From clinical symptoms to pathomechanisms
Taking NRF2 as a model in chronic diseases brings BenBedPhar to a new approach to medicine: mechanism-based disease definitions, precision diagnosis and curative therapy. This new concept is illustrated in a book, which is to be released by Springer Nature in April 2022, explaining why such an approach is badly needed.
Medicine’s achievements since 1900 are mainly due to preventing and treating infectious diseases. Other than that, medical innovation is coming to a stand-still with costs due to false incentives are increasing. This also includes biomedical research, which focuses mainly on (high) impact papers and research grants but does not measure achieved patient benefit. The biggest conceptual roadblock though is our symptom- and organ-based disease definitions perpetuated in clinical specialization and organ-based research discipline.
The book is entitled “The end of medicine as we know it.” So, not the end of medicine, only the beginning of a new one, systems medicine, which re-integrates all the organs into one big data approach. Current descriptive disease terms are endotyped according to causal molecular mechanism and treated ideally by repurposed drugs to accelerate clinical translation. Gene therapy, even germline based, will gradually eliminate rare, monogenetic diseases. Medical decision making, diagnostic and therapeutic are at least facilitated if not replaced by machine learning algorithms making fewer mistakes than medical doctors. Instead of repairing endstages of chronic diseases, the focus will be directed to prevention, leading to an entirely new wellbeing industry in which classical medicine plays a rather small part only as lifestyle coaching and prevention are the rule. From a sick care to a true healthcare system.

Two special issues about NRF2 open for submission
Currently, there are two special issues ongoing, available for the participants of the COST Action CA20121, Bench to Bedside Transition for Pharmacological regulation of NRF2 in non-communicable diseases (BenBedPhar).
The special issue entitled “Bench to bedside transition for pharmacological regulation of NRF2 in non-communicable diseases“, edited by Prof. Antonio Cuadrado (Autonomous University of Madrid, Spain), Prof. Albena Dinkova-Kostova (University of Dundee, UK), and Prof. Giovanni E. Mann (King’s College London, UK), is offered by the journal Free Radical Biology & Medicine published by Elsevier (impact factor 7.376). This Special Issue aims at integrating and disseminating novel advances in NRF2 knowledge and provides translational insights into the use of selective drugs and tools related to NRF2 pharmacology. It will contain invited review articles and primary papers from members of the BenBedPhar network, and will therefore represent an excellent collection of basic and clinical sciences in the field of NRF2 and related redox medicine issues. The deadline for the manuscript submission is June 1st, 2022. Bench to bedside transition for pharmacological regulation of NRF2 in non-communicable diseases’ – Call for papers – Free Radical Biology & Medicine – Journal – Elsevier

The special issue entitled “Transcription Factor Nrf2“, edited by Prof. Dr. Antonio Cuadrado (Autonomous University of Madrid, Spain), is open for submission in the Antioxidants journal published by MDPI (impact factor 6.313). This issue aims to present the current knowledge about the role of NRF2 in the pathophysiology of several NCDs including cancer, diabetes, autoimmune, cardiovascular, and neurodegenerative diseases, among others, as well as in the identification of NRF2 as a novel anti-inflammatory target in infectious diseases, including
Covid-19. BenBedPhar participants are invited to submit their latest research findings to this Special Issue. At this time, this special issue accounts with 9 published articles. The deadline for the submission is the 20th of June 2022.
Antioxidants | Special Issue : Transcription Factor Nrf2 (mdpi.com)

Aleksandra Buha
Science Communication Coordinator
University of Belgrade, Serbia
Hot from Pubmed
BACH1 and NRF2 in Parkinson’s disease
Disrupting the KEAP1-NRF2 pathway and consequently enhancing NRF2 activity is a promising therapeutic strategy for preventing chronic diseases underlined by low-grade redox dysregulation, such as Parkinson’s disease. However, typical NRF2 activators that target KEAP1 cysteine thiols pose the challenge of non-specific reactions with cysteine residues in a variety of cellular proteins, and may elicit important side effects. A recent paper of Ahuja et al., published in Proc. Natl. Acad. Sci. USA in November 2021, demonstrated that the levels of BACH1, a well-known nuclear transcriptional repressor of the NRF2 pathway, were found up-regulated in postmortem Parkinson’s brains and in preclinical models. The team showed that genetic deletion, and pharmacologic inhibition of BACH1 by a non-electrophilic inhibitor, was neuroprotective by up-regulating BACH1-targeted pathways, involving both Antioxidant Response Elements (ARE) and non-ARE genes. According to the obtained results the use oftheinvestigated non-electrophilic inhibitor is a promising therapeutic approach for Parkinson’s disease.
Access to the original article: https://pubmed.ncbi.nlm.nih.gov/34737234/
Astrocytes and NRF2 in Alzheimer’s disease
Alzheimer’s disease (AD) is a neurodegenerative Aß and TAU pathology. Distinct but overlapping signatures of Aß and TAU were found in human post-mortem AD astrocytes that may be associated with both deleterious and adaptive-protective signals, depending on the context. Using animal models of β-amyloidopathy or tauopathy the study of Jiwaji et al, published in January 2022 in Nat. Commun., evidenced by TRAP-seq translatome analysis a particular astrocyte signature involving repression of bioenergetic and translation machinery, induction of inflammation, proteostasis and stress-activated NRF2 target genes. The authors emphasized that the astrocyte-specific NRF2 expression induced a reactive phenotype that recapitulated elements of the human AD proteostasis signature, accompanied by reduced Aß deposition and phospho-TAU accumulation. A distinction between tauopathy vs. ß-amyloidopathy was highlighted, with enrichment in AD risk genes and sub-threshold risk loci in β-amyloidopathy. Most important, a significant overlap in the gene expression profile of astrocytes from human and mouse brains with Aß and TAU pathology was emphasized.
Access to the original article: https://pubmed.ncbi.nlm.nih.gov/35013236/
Ferroptosis and NRF2 in amyotrophic lateral sclerosis.
The study of Wang et al, published in January 2022 in Cell Death Differ., is focused on cell death pathways mediating motor neuron demise in amyotrophic lateral sclerosis (ALS). Investigations in ALS patients confirmed previous preclinical results, showing that necroptosis was indeed dispensable for motor neuron death. is coming in the forefront. In post-mortem spinal cords of both sporadic and familial ALS patients, reduced levels of glutathione peroxidase 4 (GPX4), an anti-oxidant enzyme and central repressor of ferroptosis, were detected. GPX4 depletion and ferroptosis were linked to impaired NRF2 signaling, along with dysregulated glutathione synthesis and iron-binding proteins. Accordingly, it is suggested that the use of anti-ferroptotic strategies targeting the GPX4 pathway might be beneficial for ALS treatment.
Access to the original article: https://pubmed.ncbi.nlm.nih.gov/34857917/
Vitamin C/DNA aptamer complex and NRF2 in the context of aging-related cognitive impairment and oxidative stress.
Considering that aging-related cognitive impairment is known to be associated with increased oxidative damage in the brain, there is interest in developing efficient synthetic antioxidant molecules, improving existent antioxidants such as vitamin C. A recent preclinical study published in Antioxidants inJanuary 2022 by Lee et al. showed that treatment with the new vitamin C/DNA aptamer complex (NXP032), designed for improving vitamin C stability, was able to alleviate cognitive impairment, neuronal damage, microglia activity and oxidative stress in 17 month-old female mice treated daily with NXP032 for eight weeks. The authors demonstrated that this beneficial effect of the drug complex was related to the activation of the NRF2 signaling pathway leading to increased expression of the endogenous SOD1 and GSTO1/2. Therefore, in addition to a possible direct antioxidant effect of this compound, it also induces the endogenous responses through NRF2 activation.
Access to the original article: https://pubmed.ncbi.nlm.nih.gov/35052634/
Denatonium benzoate, intestinal bitter taste receptors and NRF2.
The clinical study of Liszt et al, published in the J. Clin. Invest. in November 2021, evidenced that human intestinal bitter taste receptors (TAS2R) modulate innate immune responses and metabolic regulation in obesity. The expression of TAS2R43 and TAS2R10 genes is targeted by the bitter compound denatonium benzoate in Paneth cells and goblet cells. In relation to NRF2, the authors showed for the first time that denatonium benzoate strongly activates NRF2-mediated antioxidant and unfolded protein responses, two pathways involved in the mitohormetic response that controls aging and metabolic stress.
Access to the original article: https://pubmed.ncbi.nlm.nih.gov/34784295/
Dimethyl itaconate and NRF2 in macrophage death by pyroptosis.
The recent study of Huang et al., that was published in November 2021 in BMC Immunol., evidenced that dimethyl itaconate, an analog of itaconic acid with anti-inflammatory action, can protect against pyroptosis, a form of macrophage death found in sepsis, psoriasis and osteoarthritis. The effect is mediated by an increased antioxidant response, and involves the cytoprotective activity of NRF2. The study highlights the importance of finding novel therapeutic approaches that address pyroptosis, a promising therapeutic target being NRF2.
Access to the original article: https://pubmed.ncbi.nlm.nih.gov/34749650/
Inhibitors of KEAP1-PGAM5 protein-protein interaction as pharmacological regulators of mitophagy.
It is known that excessive ROS production induces mitochondrial depolarization, leading to the activation of PINK1-mediated Parkin translocation to mitochondria and to mitophagy, but the mechanism remains however unclear. The article of Zeb et al., published in November 2021 in Redox Biol., demonstrated that moderate production of mitochondrial superoxide and hydrogen peroxide oxidizes critical cysteine residues in KEAP1. This breaks the interaction between KEAP1 and PGAM5, leading to its accumulation due to inhibition of proteasomal degradation. This interferes with PINK1 processing, consequently stabilizing PINK1 in the mitochondrial membrane, stimulating Parkin translocation to mitochondria, and hence facilitating mitophagy and removal of excessive ROS-producing mitochondria. The authors also demonstrated that inhibitors of KEAP1-PGAM5 protein-protein interaction mimic the effect of ROS and sensitize mitochondria to autophagy, suggesting that these inhibitors are promising pharmacological regulators of mitophagy.
Access to the original article: https://pubmed.ncbi.nlm.nih.gov/34801863/
Metformin and NRF2 in diabetic vascular complications.
The study of Wang et al., published in Cell Death Dis. in January 2022, broaden our understanding of the mechanisms by which the antidiabetic drug Metformin regulates apoptosis induced by methylglyoxal in diabetic vascular complications. The authors reported that Metformin suppressed oxidative stress, prevented apoptosis and associated biochemical changes, and attenuated mitochondrial morphological alterations induced by methylglyoxal in HUVEC. The antioxidant action of Metformin was associated with increased signaling through the redox-sensitive PI3K/AKT and NRF2/HO-1 pathways, leading to markedly elevated levels of downstream antioxidants. The involvement of PI3K and NRF2 was further confirmed by the significant attenuation of the Metformin effect when specific inhibitors were used. Altogether, the study highlights at preclinical level potential application of Metformin for the treatment of diabetic vascular complications and the NRF2-mediated mechanism of action.
Access to the original article: https://pubmed.ncbi.nlm.nih.gov/35013107/
The drug-tolerant persistent state and NRF2 addiction in cancer therapeutics.
A major drawback in cancer therapeutics is related to intrinsic multidrug resistance that is associated in drug-tolerant persistent (DTP) state. The study of Zhang et al., published in January 2022 in EMBO Mol. Med., showed that DTP is underlined by increased uptake of vitamin E and cholesterol mediated by the NPC1-like intracellular cholesterol transporter 1 (NPC1L1), and NPC1L1 inhibition triggers lipid peroxides accumulation and consequent lipotoxicity. The authors demonstrated that NRF2 transcriptionally regulates NPC1L1 for counteracting the therapy-induced oxidative damage, leading to tumor recurrence, and point out the importance of disrupting the NRF2-KEAP1 axis in the context of cancer therapy, for overcoming the DTP-mediated resistance of cancer cells.
Access to the original article: https://pubmed.ncbi.nlm.nih.gov/35023619/
Gina Manda
Vice-Chair
Elena Milanesi
Gender Equality Officer
Victor Babes
National Institute of Pathology, Romania

